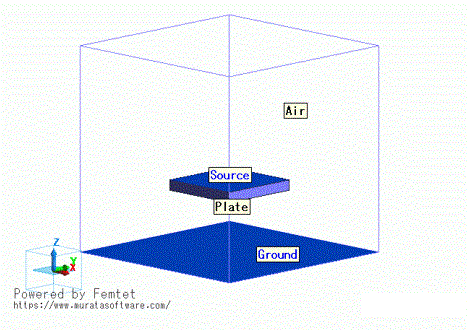
Home / Examples / Fluid Analysis [Bernoulli] / Example 19: Convection Caused by Generation of Impurity Substance
The diffusion analysis with the weight of the diffusing materials taken into account is applied for the convection caused by an impurity substance generated in a closed space.
The resulting convection and concentration distribution are compared between cases where the diffusing material is water vapor (H2O), which is lighter than air, and cases where it is sulfur dioxide (SO2), which is heavier.
The flow velocity distribution, the flow velocity vectors, and the impurity substance concentration distribution are solved.
Unless specified in the list below, the default conditions are applied.
Results will vary depending on Femtet version and the PC environment.
Item |
Settings |
Analysis Space |
3D |
Model Unit |
m |
Item |
Tab |
Settings |
||||||||
Solver |
Solver |
Fluid Analysis [Bernoulli] |
||||||||
Analysis Type |
Fluid Analysis |
Transient Analysis |
||||||||
Timestep |
Transient Analysis |
Timestep: Specify
|
||||||||
Diffusion Analysis |
Fluid Analysis Diffusion Analysis Setting |
Select Execute diffusion analysis Analysis Type: Transient Analysis Type and Unit of Diffusion Quantity: Molar Concentration [mol/m3]
Diffusing Material Setting:
|
||||||||
Meshing Setup |
Mesh |
General Mesh Size: 10 [mm] |

Body Number/Type |
Body Attribute Name |
Material Name |
0/Solid |
Air |
000_Air * |
1/Solid |
Plate |
001_Alumina * |
* Available from the material DB
Boundary Condition Name/Topology |
Tab |
Boundary Condition Type |
Settings |
Ground/Face |
Fluid |
Solid Wall |
|
Source/Face |
Fluid |
Solid Wall |
Diffusion Analysis Setting Select Flux Molar Concentration Flux: 0.01 [mol/m2/s] |
Outer Boundary Condition |
Fluid |
Solid Wall |
|
The flow velocity vectors on the section at y=0 at 20 [s] are shown below.
When water vapor, which is lighter than air, is generated on the top of Plate, an upward flow occurs.
When sulfur dioxide, which is heavier than air, is generated on the top of Plate, a downward flow occurs.
H2O-Wall |
SO2-Wall |
|
|
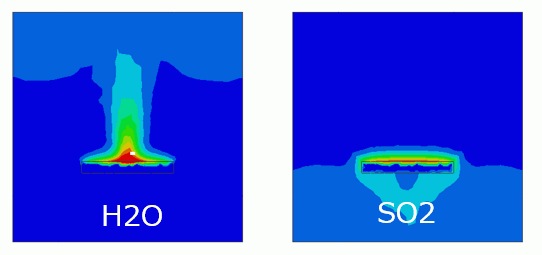
The contours of molar concentration on the section at y=2 at 5 [s], 10 [s] , and 20 [s] are shown below.
If the impurity substance is water vapor, the substance stays at the top of the closed space, and if sulfur dioxide, stays at the bottom.
Time |
H2O-Wall |
SO2-Wall |
5 [s] |
|
|
10 [s] |
|
|
20 [s] |
|
|