Home / Examples / Electric Analysis [Coulomb] / Example 9 Plating (Plating a Device in the Bath)

A device is immersed in the plating bath and plated. The plating is analyzed.
By modeling the anode and cathode, the distribution of electric potential at the electrodes can be taken into account.
How to create the model using a plating wall is explained at the end of this example.
Unless specified in the list below, the default conditions will be applied.
Results will vary depending on Femtet version and the PC environment.
Item |
Settings |
Analysis Space |
3D |
Model Unit |
mm |
The material is conductor for the plating analysis.
Select the [Perform the plating analysis] option.
Item |
Settings |
Solver |
Electric Analysis [Coulomb] |
Analysis Type |
Static Analysis (Resistance) |
Material |
Conductor |
Options |
Select Perform the plating analysis |
Meshing Setup |
General Mesh Size: 6.0 [mm]*1
Meshing Control |
Matrix Solver Type |
Direct Method*3 |
*1: Mesh size of 6.0 mm is set as the simulation accuracy will not be enough with the automatically-set mesh size of 12.0 mm.
*2: By this setting, the curved face can be meshed more smoothly.
See [Meshing Control] for the detail.
* 3: In the electric field analysis, if Automatic is selected for the Matrix Solver Type, the iterative method is used.
In this example, Direct Method is selected as it converges faster than the iterative method.
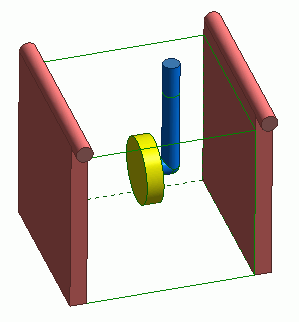
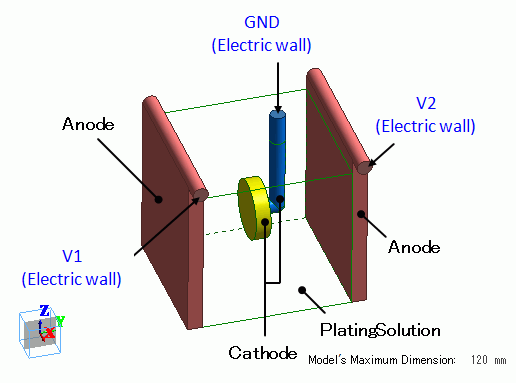
Box body (green) is the plating solution. As the target object for plating, a disc part (yellow) is placed in the box.
The disc part is a cathode. Anodes (red) are placed on both sides of the disc part.
Assuming that the protruding portion of the cylinder on top of the anodes are connected to the current power source, electric walls V1 and V2 are set on the anodes.
The disc part is supported by a conductive supporting body (blue) that flows current.
Assuming the end of the conductive part is connected to the ground, the electric wall of ground, GND, is set on the end.
Create solid bodies for the plating bath, the anodes, the disc part (cathode), and supporter of the disc part (conductor). Body attributes are plating solution and metal.
Body Number/Type |
Body Attribute Name |
Material Name |
3/Solid |
Cathode |
Metal |
9/Solid |
Anode |
Metal |
10/Solid |
Anode |
Metal |
11/Solid |
Cathode |
Metal |
12/Solid |
Plating Solution |
Solution |
The body attribute is set as follows.
Body Attribute Name |
Plating Tab |
||||||||||||||
Anode |
Type of Plating Body: Anode
Overvoltage Type: Linear
|
||||||||||||||
Cathode |
Type of Plating Body: Cathode
Overvoltage Type: Tafel's Law
|
||||||||||||||
Plating Solution |
Type of Plating Body: Plating Solution
|
||||||||||||||
The material properties are set as follows.
Material Name |
Conductivity Tab |
Metal |
Conductivity Type: Conductor Conductivity: 10 x106 [S/m]
|
Solution |
Conductivity Type: Conductor Conductivity: 10 [S/m]
|
The boundary conditions are set as follows.
Boundary Condition Name/Topology |
Tab |
Boundary Condition Type |
Settings |
Ground/Face |
Electric |
Electric Wall |
Ground |
V1/Face |
Electric |
Electric Wall |
Current 1 A |
V2/Face |
Electric |
Electric Wall |
Current 1 A |
Current density of the plating solution is shown in Fig. 1.
Bodies except the plating solution are hidden
because current density of the plating solution cannot be seen due to the current density of electrodes if the electrode bodies are shown.
The current from the anodes is observed to flow to the cathode which consists of the disc part and the supporting body.
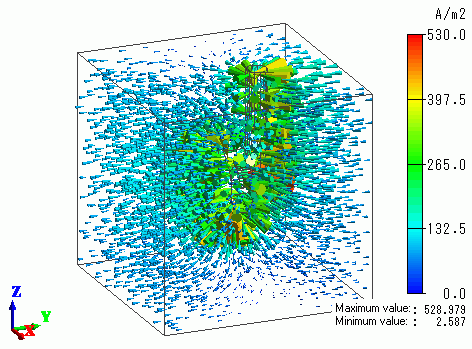
Fig. 1 Distribution of current density in plating solution
Between the bodies with attributes of the electrodes (anode and cathode) and the body with the attribute of plating solution,
a body with an attribute name of RESEVED_dual is automatically created.
The vector field (electric field or current density) of this body is extraordinary.
Hide the body with the attribute name of RESERVED_dual
before observing that field.
Fig. 2 shows the current density (normal component) on the disc part's surface and
Fig. 3 shows the plating thickness extracted on the disc part, each in the form of contours. Only the disc part body is shown in both figures.
Both parts are placed such that the supporting body is on your side.
The supporting body is directed upward from the disc's center.
In Fig. 2, you can see the current density at the upper area of the part is small due to the absence of current by obstruction from the supporting body.
Fig. 3 shows that the plating thickness is distributed according to the current density distribution.
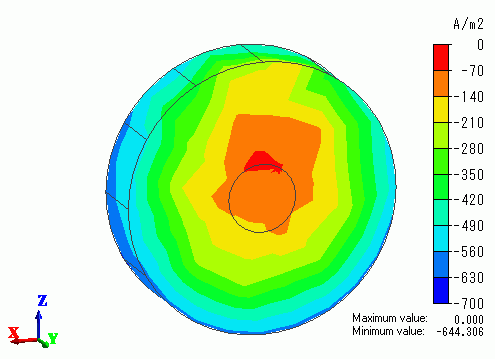
Fig. 2 Current density (normal component) on the surface of part 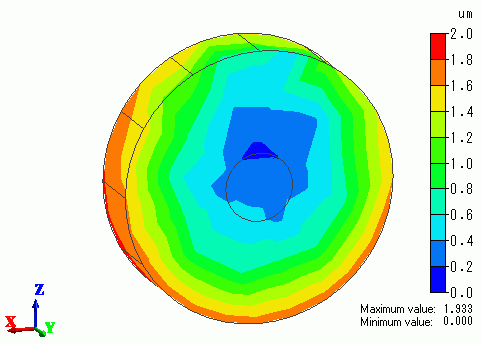
Fig. 3 Plating thickness extracted on the part
Fig.4 shows the contour of the electric potential on one of the anodes.
You can see that there is electric potential distribution in the anode and the distribution is taken into account in the analysis.
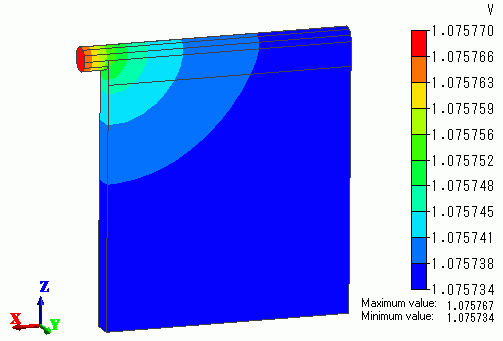
Fig. 4 Electric potential distribution of the anode
In this example, the bodies are created for the anode and cathode.
This method has the advantage that electric potential distribution in the electrodes can be taken into account.
It has, however, a disadvantage that the number of meshes increases according to the number of electrodes, and a longer time is required for calculation.
If the plating wall boundary condition is used instead of the electrode body,
it can reduce meshes and shorten calculation time while the electric potential distribution in the electrode cannot be taken into account.
Fig. 5 shows the model with the plating wall replacing the electric wall.
The plating solution body is cut out and hollow where the disc part is supposed to be.
