Home / Technical Notes / Fluid Analysis/Fluid-Thermal Analysis / Diffusion Analysis
Diffusion Analysis
Diffusion analysis of the fluid solver is explained.
For the details of the setup, please refer to [Fluid (Fluid-Thermal)] tab.
1. Diffusion Analysis
The diffusion of materials in fluid can be analyzed.
Diffusion by the difference in concentration of materials and advection under which materials flow in a fluid are taken into account.
In turbulent analysis, the increased diffusion by turbulence is taken into account.
・Water vapor diffusion in air
・Toxic material diffusion in air
・Concentration of nitrogen in air
can be solved.
The analysis is based on the precondition that, the concentration of the diffusing material does not affect the flow of main components.
Therefore, some problems may not be solved.
Phase transition, such as from water vapor to water, cannot be solved in this analysis.
Efficiency of ventilation can be estimated in this analysis.
Refer to the tutorial Fluid Analysis 2 (Bernoulli, Diffusion Analysis: Automatic Internal Fluid Creation) for how to conduct the diffusion analysis.
1.1 Type and Unit of Diffusion Quantity Setting
The concentration of diffusing material is calculated.
The concentration is defined as molar concentration [mol/m3], mole fraction, mass concentration [kg/m3], or mass fraction.
In the Diffusion Analysis Setting of the analysis setting, a type of diffusion quantity is selected from the eight options shown below.
・No unit
・Molar concentration [mol/m3]
・Mass Concentration [kg/m3]
・Mole fraction (no unit)
・Mole fraction [%]
・Mass fraction (no unit)
・Mass fraction [%]
・Low Concentration [ppm]
1.2 Diffusion Quantity Setting
The value of diffusion quantity is set at the following settings for analysis.
・Analysis Condition > Diffusion Analysis Setting: Ambient value (default)
・Analysis Condition > Diffusion Analysis Setting: Initial value in transient analysis
・Body attribute > Diffusion Analysis Setting: Initial value in transient analysis for each body attribute
・Boundary Condition > Diffusion Analysis Setting [Inflow]: Value of diffusion quantity that comes in from the inlet boundary
・Boundary Condition > Diffusion Analysis Setting [Wall Face]: Values on the wall face, Inflow/Outflow flux on the wall face, Ambient value and transfer coefficient on the outside of the wall
Examples
(1) Air of 30% humidity flows into air of 60% humidity.
The type of diffusing quantity is mole fraction [%].
The main component is dry air (humidity 0%) and the ambient value is 0%.
The initial value for transient analysis is 30 [%].
The Inflow value of the inlet boundary is 60 [%] .
*The method is not applicable if the weight of diffusing materials is taken into account. Try the (2) or the (3) method instead.
(2) Calculate (1) in molar concentration.
The type of diffusion quantity is molar concentration [mol/m3].
To convert humidity to molar concentration, the amount of saturated water vapor is first calculated.
The amount of saturated water vapor is converted to molar concentration with the the equation of state.
The Tetens equation can estimate an amount of saturated water vapor at a specified temperature.

p: Saturated water vapor pressure, T: Temperature [deg]
The amount of saturated water vapor is converted to molar concentration with the equation of state.

C: Molar concentration of saturated water vapor [mol/m3], R: gas constant of 8.314462618 [J/K/mol], Temperature: T [K]
When the temperature is 25 [deg], the molar concentration of saturated water vapor is 1.278 [mol/m3].
When the humidity is 30%, the molar concentration of water vapor is 1.278 x 0.3 = 0.3834 [mol/m3].
When the humidity is 60%, the molar concentration of water vapor is 1.278 x 0.6 = 0.7668 [mol/m3].
The main component is dry air (humidity 0%) and the ambient value is 0[mol/m3].
The initial value for transient analysis is 0.3834 [mol/m3].
The Inflow value of the inlet boundary is 0.7668 [mol/m3].
(3) Calculate (1),(2) in mass concentration of water vapor.
The type of diffusing quantity is mass concentration [kg/m3].
The molar concentration of saturated water vapor obtained in (2) is converted to mass concentration with the molecular weight.
The molecular weight of water (H20) is 18 [g/mol].
When the humidity is 30%, the molar concentration of water vapor is 0.3834 * 18 * 0.001 = 0.006901 [mol/m3].
When the humidity is 60%, the molar concentration of water vapor is 0.7668 * 18 * 0.001 = 0.013802 [mol/m3].
The main component is dry air (humidity 0%) and the ambient value is 0 [kg/m3].
The initial value for transient analysis is 0.006901 [kg/m3].
The inflow value of the inlet boundary is 0.013802 [kg/m3] .
(4) Nitrogen gas flows into air.
The type of diffusing quantity is mole fraction [%].
As the main component is air of about 80% nitrogen, the ambient value is set to 80 [%].
The Inflow value of the inlet boundary is 100 [%] .
2.Take into Account Buoyancy.
2.1 Buoyancy from Weight and Concentration of Diffusing Materials
The analysis is based on the precondition that the concentration of the diffusing material does not affect the flow of main components.
The fluid containing the diffusing material can change in average density and average viscosity depending on the concentration of the diffusing material.
High concentration may affect the flow.
Close attention should be paid to spatial change in the average density of a diffusing material. That change can generate buoyancy and then natural convection flow.
If a diffusing material is lighter than the main component of the fluid, the diffusing material tends to stay around the top due to buoyancy.
On the other hand, if a diffusing material is heavier, the diffusing material tends to stay around the bottom.
The "Lighter" means the molar mass (molecular weight) of the diffusing material is smaller than that of the main component of the fluid.
The "Heavier" means the molar mass (molecular weight) of the diffusing material is larger than that of the main component of the fluid.
For gas, the magnitude of buoyancy is defined as follows.

f: Buoyancy Vector [N/m3]
M: Molar mass (Molecular weight) of diffusing material [kg/mol]
Mref: Molar mass (Molecular weight) of main component of fluid
C: Molar concentration difference of diffusing material [mol/m3]
g: Gravity acceleration vector [m/s2]
The diffusing material is water vapor.
・Molecular weight of water: 18 [g/mol]
・Molecular weight of air (the main component of fluid): 28.96 [g/mol]
Water vapor is lighter than dry air and the resultant buoyant vector is in the opposite direction from the gravity acceleration vector. Air containing more water vapor generates more buoyancy.
For liquid, the magnitude of buoyancy is defined as follows.
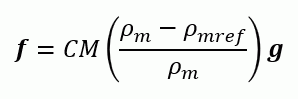
ρm: Density of diffusing material [kg/m3]
ρmref: Density of main component of fluid [kg/m3]
2.2 Take into Account Weight of Diffusing Materials
Select [Take into account weight of diffusing materials] in [Diffusion Analysis Setting].
Select a diffusing material in [Diffusing Material Setting] and set its molar mass.
See [Differential Equations in Fluid Analysis/Fluid-Thermal Analysis ] for details about equations.
2.3 Determine If Buoyancy Should Be Taken into Account
Whether the weight of diffusing materials needs to be taken into account depends on which flow is dominant by forced convection or buoyancy.
If forced convection affects more than natural convection, buoyancy needs not be taken into account.
The following explains how the ratio of Reynolds number and Grashof number will determine whether buoyancy needs to be taken into account or not.
・Reynolds number

Re: Reynolds number
U:Typical flow velocity [m/s]
L: Typical length
ν: Kinematic viscosity [m2/s]
This is an indicator that shows the balance between inertial and viscous forces. Reynolds number is bigger than 1 if inertial force is more dominant.
・Grashof number

Gr: Grashof number
g: Gravity acceleration [m/s2]
βc:Coefficient of volumetric thermal expansion [m3/mol]
C: Expected molar concentration difference [mol/m3]
L: Typical length
ν: Kinematic viscosity [m2/s]
This indicates the balance between buoyancy and viscous forces. It is bigger than 1 if buoyancy is more dominant.
Coefficient of volumetric thermal expansion is defined as follow.

Mref: Molecular weight of main component of fluid [kg/mol]
M: Molecular weight of diffusing material [kg/mol]
ρref: Density of fluid main component [kg/m3]
・The ratio of Grashof number and Reynolds number (also called Richardson number)

Gr: Grashof number
Re: Reynolds number
g: Gravity acceleration [m/s2]
βc:Coefficient of volumetric thermal expansion [m3/mol]
C: Expected molar concentration difference [mol/m3]
L: Typical length
U:Typical flow velocity [m/s]
This is the indicator that shows the balance between inertial force and buoyancy. It is smaller than 1 if inertial force is more dominant.
In the mixed flow of forced convection and natural convection by buoyancy, if the indicator is smaller than 1, forced convection is dominant,
and if it is larger than 1, natural convection is dominant.
If the indicator is smaller than 1, natural convection is less dominant and buoyancy needs not be taken into account.
2.4 Tool to Determine If Buoyancy Should Be Taken into Account
The Excel tool is available for the calculation described in 2.3.
・Viscosity, density, molecular weight of main component of fluid
・Molecular weight of diffusing material
・Typical length (flow velocity)
・Typical length (dimensions)
・Expected molar concentration difference of diffusing material
Use the tool as needed.
CheckApplicableOfDiffusionAnalysis.xlsx
2.5 Examples of Determination of Application Range
2.5.1 Diffusion of Water Vapor
It is determined whether buoyancy should be taken into account in the case of saturate air (100% humidity) diffusion with typical fluid velocity of 1 m/s and typical length of 1m.
Calculation is performed assuming that humidity changes from 0% to 100% in the analysis domain.
1. Molar concentration of saturated water vapor at 25 deg C is obtained with the "Water vapor" sheet of Excel.
The molar concentration of 1.28 [mol/m3] is given.
2. Enter typical flow velocity of 1 m/s, typical length of 1m, molecular weight of diffusing material of 18 [g/mol], and molar concentration difference of diffusing material of 1.28 [mol/m3] in the "Determination" sheet of Excel.
The ratio of Grashof number and Reynolds number is 0.12.
As forced convection is dominant, diffusion analysis is applicable.
With higher temperatures, molar concentration of saturated water vapor will increase. In such a case, Grashof and Reynolds numbers will increase, and the diffusion analysis may not be applicable.
2.5.2 Diffusion of Nitrogen Gas
It is determined whether buoyancy should be taken into account in the case of diffusion of nitrogen gas with typical velocity of 1 m/s and typical length of 10m.
Calculation is performed assuming that nitrogen mole fraction changes from 80% to 100% in the analysis domain.
1. In the "Equation of state" sheet of Excel, under pressure of 1e5 [Pa] and temperature of 25 [deg C], molar concentration is obtained at mole fraction of 80% and 100%.
Molar concentration at mole fraction of 80% and 100% are 32.3 [mol/m3], and 40.3 [mol/m3], respectively.
The molar concentration change in the analysis domain is 40.3 - 32.3 = 8.0 [mol/m3].
2. In the "Determination" sheet of Excel, enter typical flow velocity of 1 m/s, typical length of 10m, molecular weight of diffusing material of 28 [g/mol], and molar concentration difference of diffusing material of 8.0 [mol/m3].
The ratio of Grashof number and Reynolds number is 0.66.
As forced convection is dominant, diffusion analysis is applicable.
3. Calculation of Ventilation Efficiency
Diffusion analysis solves ventilation.
Typically, ventilation in the room with a ventilator is analyzed.
2D model is applied.
For 3D analysis, see Example 12 Room Ventilation, Example 13 Room Ventilation (Air Age Calculation) for more information.
The flow rate of the ventilator is set to 15 [m3/min] for one circulation per 1 [min].
The room volume is 15 [m3 ](6m x 2.5m x 1m in depth) . This will lead to one cycle circulation for 1 [min] at the flow rate of 15 [m3/min].
Assuming that the door is open, set natural inflow/outflow to the door.

Streamlines are shown as below. They are extending from the door to the ventilator.

3.1 Air Replacement Analysis with Transient Analysis
Select [No unit].
The transient analysis is performed with the initial value of 1 and the ambient value of 0.
The area having the value of 1 is where old air remains and the area having the value of 0 is where new air has replaced old air.
The area where the value keeps 1 for a long time is where stagnation occurs and ventilation hardly occurs.
|
|
Contour Diagram |
|
Time:20 [s] |
|
|
Time:40 [s] |
|
|
Time:60 [s] |
|
3.2 Analysis of Air Age, Air Residual Lifetime, and Air Lifetime
Air age, air residual lifetime, and air lifetime are used to evaluate ventilation.
Air age, air residual lifetime, and air lifetime have an unit of time [s]. For evaluation, they can be normalized with the number of ventilations.
The number of ventilations is calculated by (ventilation flow rate)/(ventilation volume), which means how many times ventilation can be performed in 1 [s].
If circulation is performed one time per minute, the number of ventilations per second is 1/60.
Air Age
The air age is the time required for air to travel from the inflow port to a specific port.
A closer port to the inflow port, where new air is easily reached, has shorter air age.

Air age is 0 [s] near the door and about 60[s] near the ventilator.
The ventilation volume is set such that air circulates once per 1 [min]. Therefore, the result is considered reasonable.
Air Residual Lifetime
The time required for air to travel from a specific port to the outflow port
A closer port to the outflow port, where old air is easily discharged, has shorter air residual lifetime.

Air age is 60 [s] near the door and about 0[s] near the ventilator.
The ventilation volume is set such that air circulates once per 1 [min]. Therefore, the result is considered reasonable.
Air Lifetime
The air lifetime is the time required for air to travel from the inflow port to the outflow port through a specific port.
(Sum of air age and air residual lifetime)
A stagnating area with less ventilation has larger value.

The areas on the corners have longer air lifetime and are stagnated.
Those areas show no streamline, which indicates air ventilation hardly occurs.





